Standardize Your Immune Assays for Your Clinical Trials
Standardization of immunoassays is a critical component for reliable and successful performance in clinical trials. Our team of experts has identified procedural and material variations affecting assay results, and we offer a comprehensive suite of tools and products to overcome these variables and help you to achieve reliable results.
These resources include:
- Highly trained technical personnel at our CRO laboratory in case we perform the work for you
- Centralized blood processing services for multicenter clinical trials in the U.S. by our CRO contract laboratory
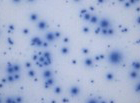
- Generation of functional PBMC (tested in ELISPOT assays) using CTL's cryopreservation reagents
- Support with logistic and training for standardized blood collection and processing at international clinical sites
- Methods, reagents and instrument for counting viable PBMC: LDA counting suite, cell counting kits etc.
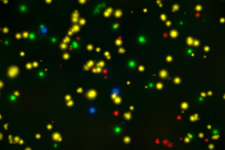
- Optimized T and B cell ELISPOT kits for enzymatic or multiplexed FluoroSpot applications
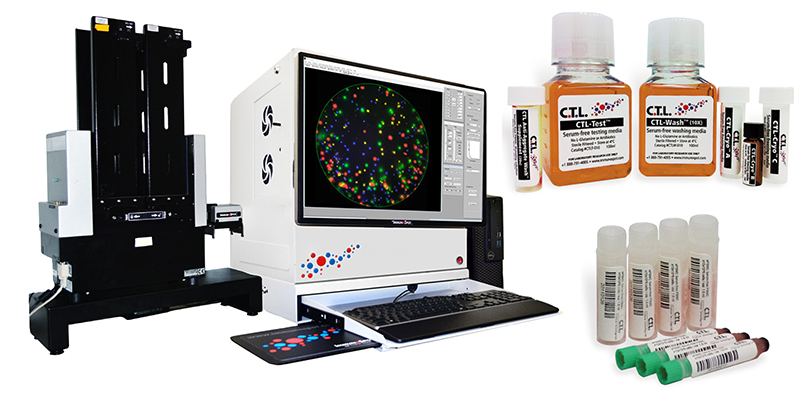
- CTL's serum-free media solutions for PBMC: thawing, washing, and testing
- Standardized positive control antigens for CD4 and CD8 specific assays
- ImmunoSpot® instrumentation for capturing images and standardized spot counting
- CTL SpotStat® and SpotMap® solutions for analyzing data
- QC packages for instrumentation and reference samples.